
Ultimately, it is the brain that controls the perception of pain and determines which stimuli are useful and which are to be ignored. Furthermore, afferent pathways interact with each other in many ways other than the “gates” mentioned above. 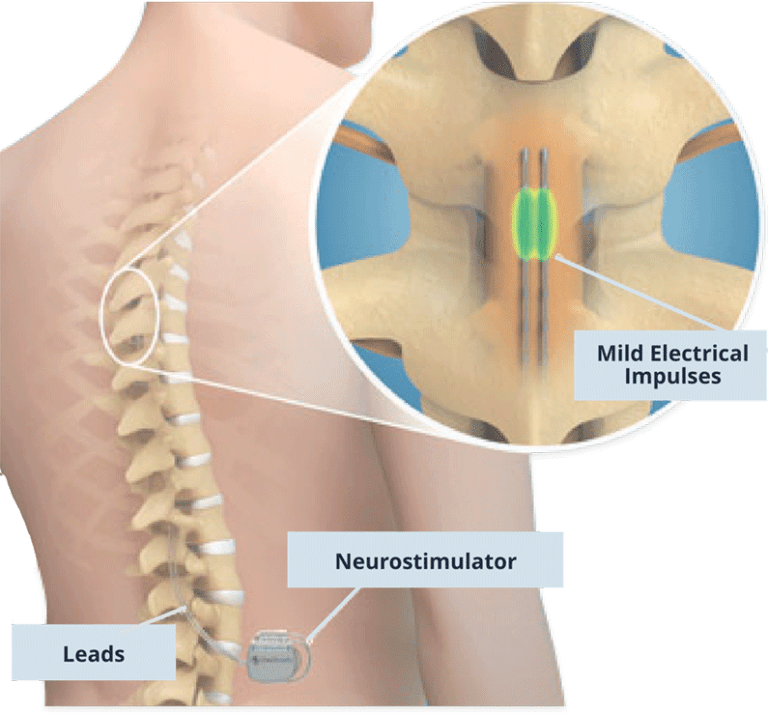
It should be noted, however, that pain perception is a product of the brain’s processing of afferent inputs the perception of pain involves numerous sensory, affective, and cognitive components. Subsequently, techniques for peripheral nerve stimulation were developed on the same principle.Īdditional theories that have attempted to explain the pain relief provided by PNS include (1) excitation failure in c-fiber nociceptors and suppression of dorsal horn activity, (2) stimulation-induced blockade of cell membrane depolarization preventing axon conduction propagation, (3) decreased hyperexcitability and long-term potentiation of dorsal horn neurons, and (4) depletion of excitatory amino acids (glutamate, aspartate) and increased release of inhibitory transmitters (GABA). The theory explains why rubbing or massaging a painful site relieves pain: it causes stimulation of nonnociceptive fibers, which close the ‘gates’. This means that pain is perceived when these physiological ‘gates’ give way to pain signals and is less intense or not perceived when the gate closes. The input from small-diameter fibers opens the “gates” and results in nociception (pain). Nonnociceptive input activates large-diameter sensory fibers that close the “gates” to nociceptive input, which is otherwise carried via small-diameter fibers. Gate control theory suggests that “gates” at the spinal cord dorsal horn laminae competitively regulate nonnociceptive and nociceptive input. The most attractive hypothesis for the mechanism underlying PNS is gate control theory, which was published in Science in 1965. Within the last decade, PNS has been shown to be effective in the treatment of several painful conditions including complex regional pain syndrome, postherpetic neuralgia, posttraumatic neuropathy, cranial neuralgias, and various headache disorders. These advancements include improvement in ultrasound technologies, integration of ultrasound into clinical practice, percutaneous implantation techniques, smaller devices, and rechargeable and larger-capacity batteries. Since then, there have been tremendous advancements in the field of neuromodulation, including peripheral nerve stimulation (PNS). After completion of the procedure, four of the eight patients experienced more than half an hour of pain relief. The stimulation consisted of 0.1ms pulses at a rate of 100 Hz for two minutes the voltage was adjusted until patients reported a tingling sensation in the affected area. Electrical stimulation was applied to eight patients with chronic neuropathic pain. The first report of pain relief following electrical stimulation comes from Wall and Sweet, in 1967. The modern era of neuromodulation for intractable and chronic pain was conceptualized after the publication of Wall and Melzack’s gate control theory of pain in 1965. The use of electricity for treatment of chronic pain can be traced back to the Romans, who used electric eels, a fresh water predator that can generate and release electricity to stun prey, for the treatment of various painful conditions. In this review, we discuss the history, pathophysiology, indications, implantation process, and outcomes of employing peripheral nerve stimulation to treat chronic pain conditions. 
Recently, a new generation of peripheral nerve stimulation devices has been developed these allow external pulse generators to transmit impulses wirelessly to the implanted electrode, and their implantation is significantly less invasive. It has been shown to be effective for chronic, intractable pain that is refractory to conventional therapies such as physical therapy, medications, transcutaneous electrical stimulations, and nerve blocks. Peripheral nerve stimulation has been used in the treatment of several chronic pain conditions including pain due to peripheral nerve dysfunctions, complex regional pain syndrome, and cranial neuralgias.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
